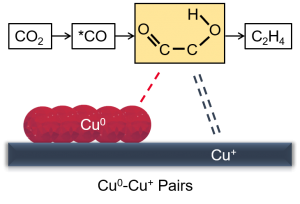
Cu‐based electrocatalysts can effectively facilitate carbon dioxide electrochemical reduction (CO2ER) to produce multi‐carbon products. However, the roles of Cu0 and Cu+ and the mechanistic understanding remain elusive. This paper describes the controllable construction of Cu0-Cu+ sites derived from the well‐dispersed cupric oxide supported on copper phyllosilicate lamella to enhance CO2ER performance. Specifically, 20% Cu/CuSiO3 shows the superior CO2ER performance with 51.8% C2H4 Faraday efficiency at ‐1.1 V vs reversible hydrogen electrode (RHE) during the 6 hours‐test. In situ attenuated total reflection infrared spectra and density functional theory calculations were employed to elucidate the reaction mechanism over Cu0-Cu+ sites. The enhancement in CO2ER activity is mainly attributed to the synergistic effect of Cu0‐Cu+ pairs: The Cu0 site activates CO2 and facilitates the following electron transfers; while the Cu+ site strengthens the *CO adsorption to further boost C‐C coupling. This paper provides an efficient strategy to rationally design Cu‐based catalysts with viable valence states to boost CO2ER.
Xintong Yuan, Sai Chen, Dongfang Cheng, Lulu Li, Wenjin Zhu, Dazhong Zhong, Zhi-Jian Zhao, Jingkun Li, Tuo Wang, and Jinlong Gong*, Controllable Cu0-Cu+ Sites for Electrocatalytic Reduction of Carbon Dioxide. Angew. Chem. Int. Ed. 2021, DOI: 10.1002/ange.202105118